What are all possible cases of hybridization of an sp2-hybridized carbon atom? How can they be identified? - Quora

How many sp2 hybridized carbon atoms are present in this molecule? I thought it was 4 but it's 9 for some reason?? : r/Mcat

A structure for a molecule that meet the following description: Contains two sp2-hybridized carbons and two sp3-hybridized carbons | Homework.Study.com

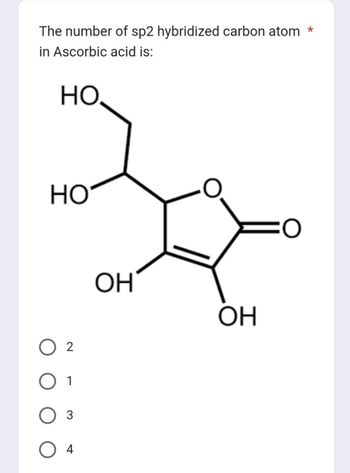

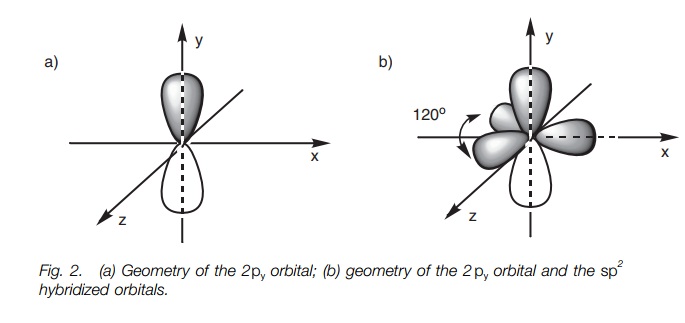
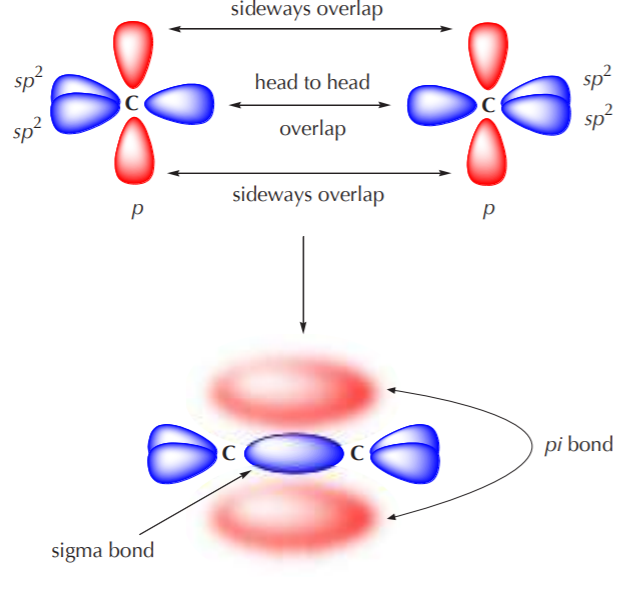

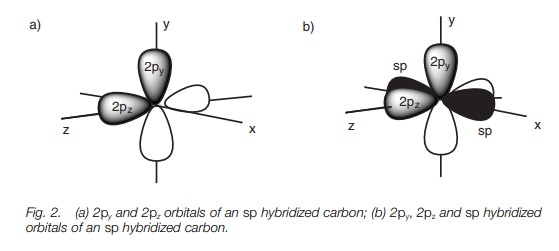
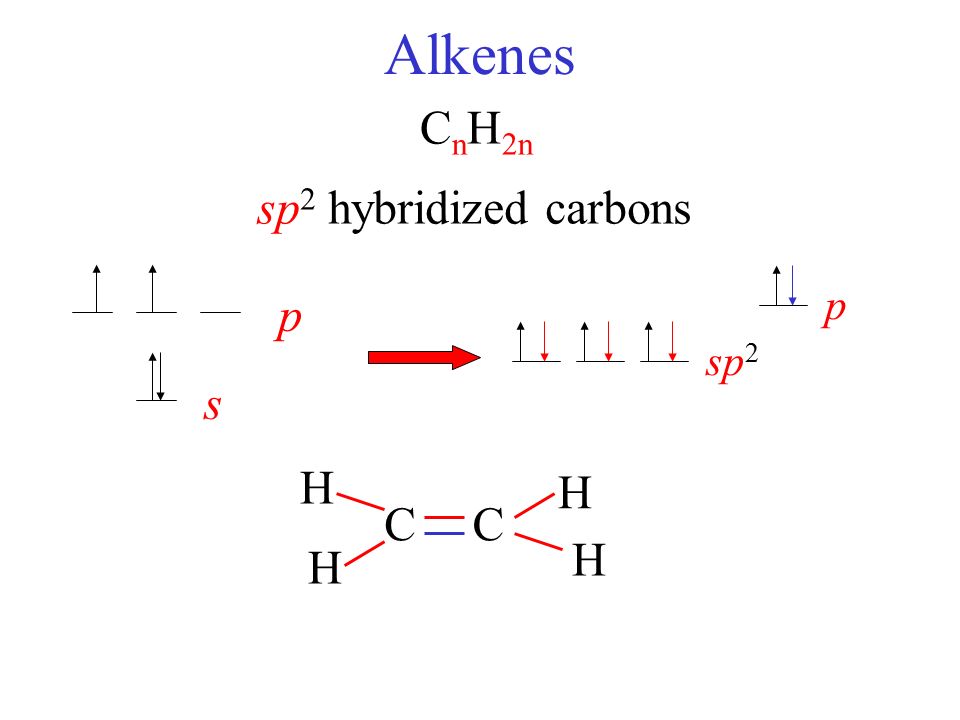


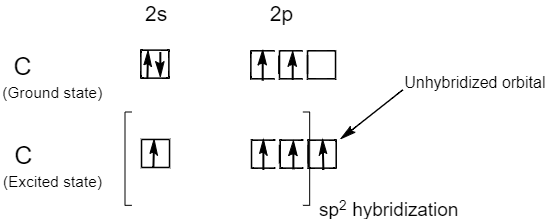

/chapter2/pages1and2/page1and2_files/sp2hyb.png)

