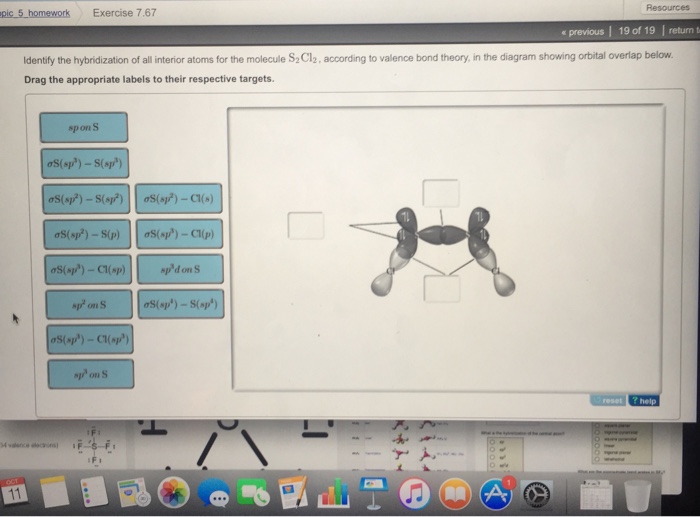
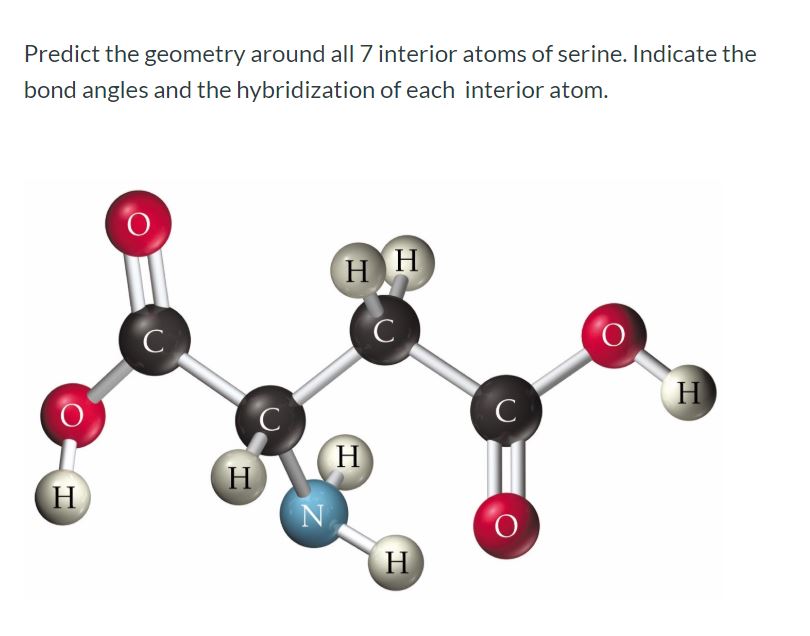
For each atom listed, identify the geometry as one of the following: tetrahedral, square planar, trigonal planar, trigonal pyramidal, or linear. | Homework.Study.com

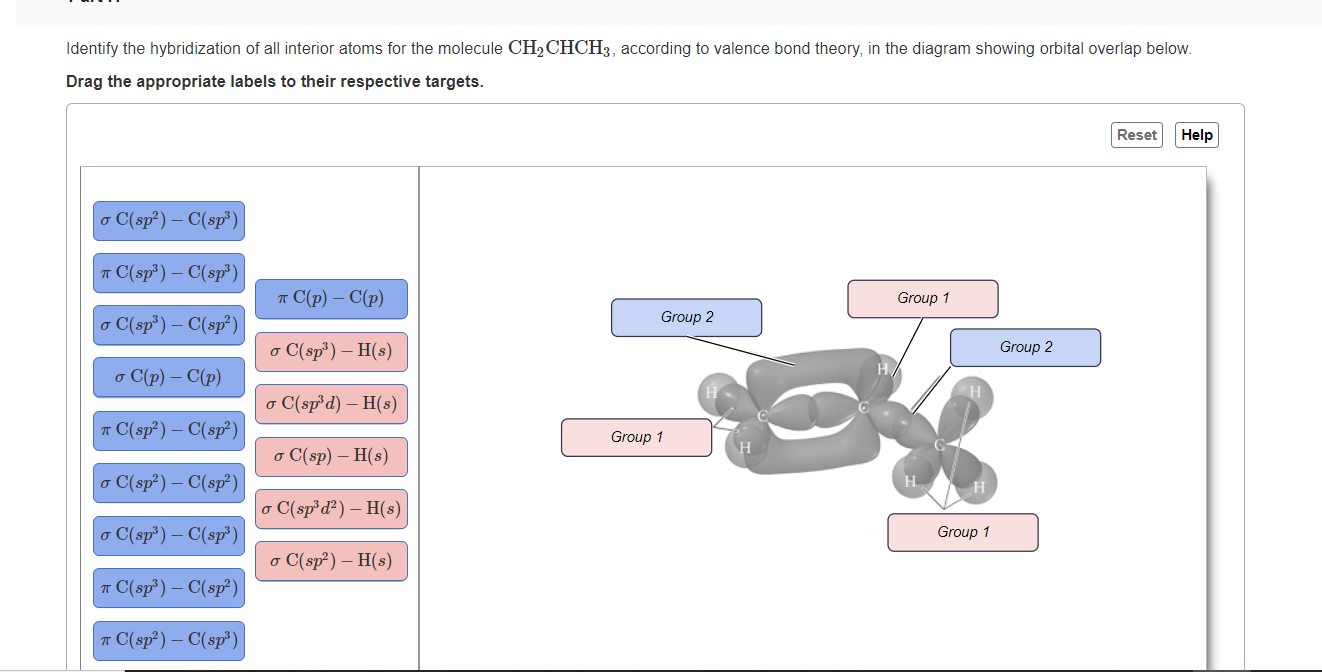
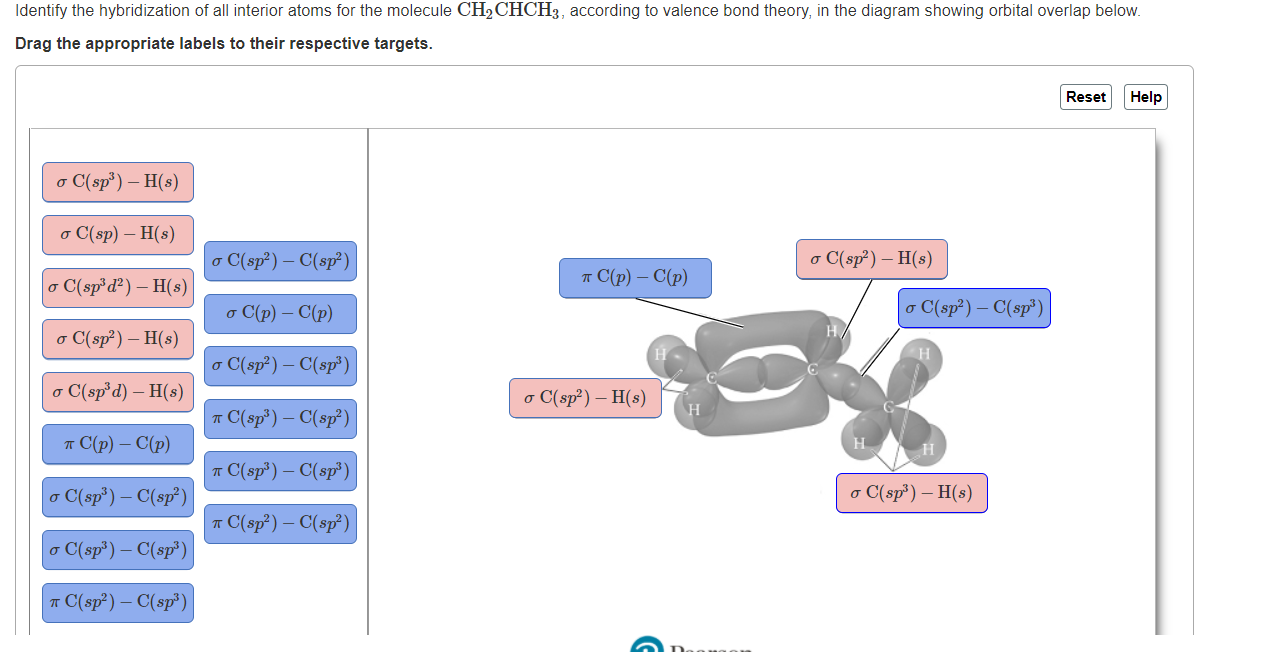
Describe the molecular geometry around each carbon atom in CH2CHCH3 using VSEPR theory. | Homework.Study.com
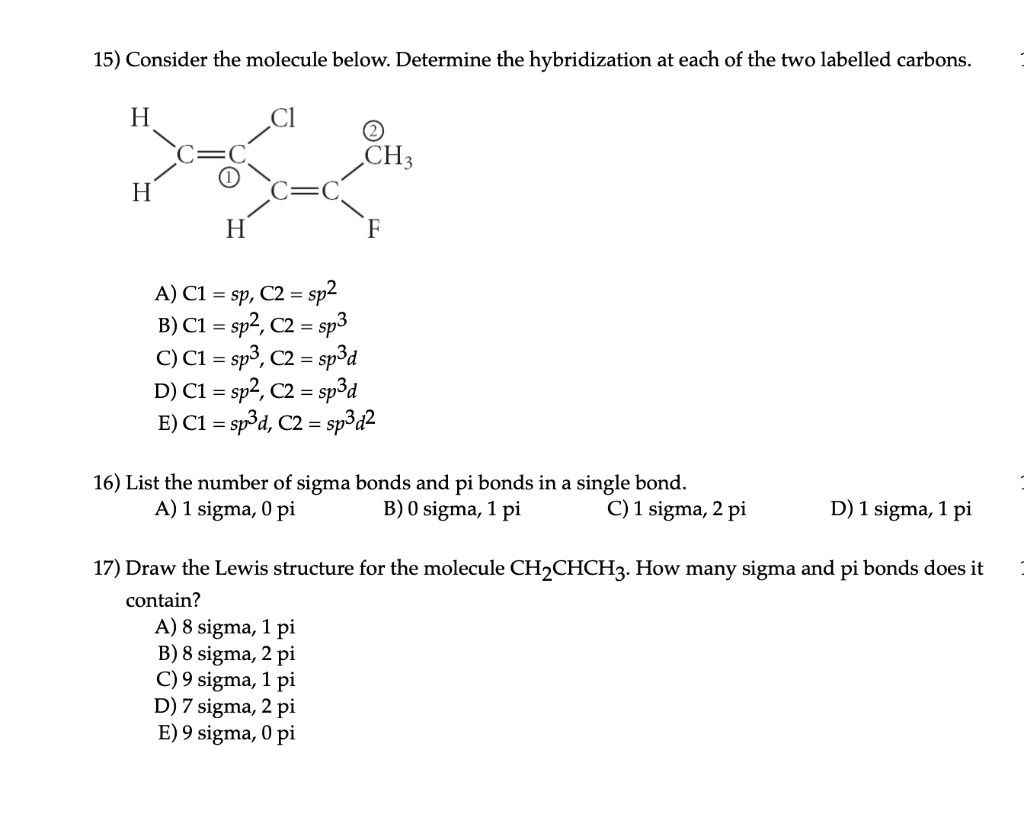
SOLVED: 15) Consider the molecule below. Determine the hybridization at each of the two labeled carbons. H 2 H H A) C1 = sp, C2 = sp2 B) C1 = sp2, C2 =
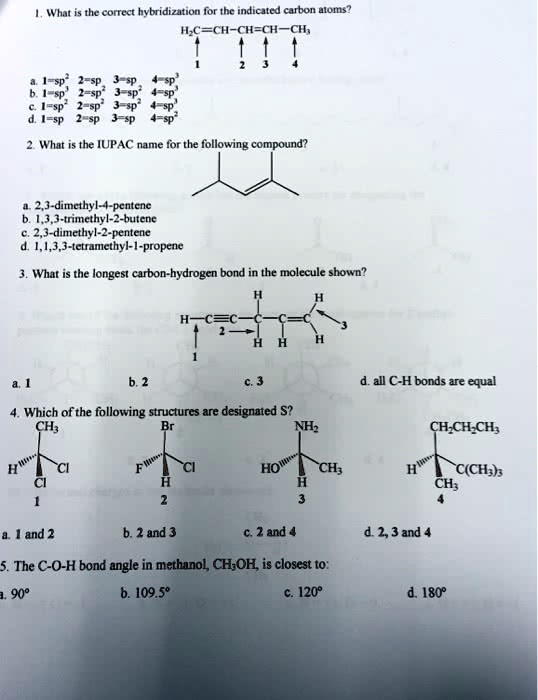
SOLVED: 1. What is the correct hybridization for the indicated carbon atoms? H2C=CH-CH=CH-CH3 a. -sp2, 3-sp3, 4-sp2. 2. What is the IUPAC name for the following compound? a. 2,3-dimethyl-4-pentene b. 1,3,3-trimethyl-2-butene c.

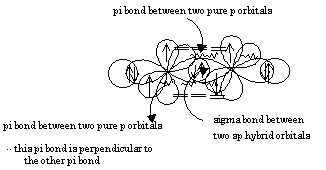
SOLVED: In valence bond theory, which orbital hybridization combination is found in O=C=O? A. 2s in carbon with a 2s in oxygen B. 2p in oxygen with a 2p in carbon C.
University of Thi-Qar … College Of Science ……….. Department of Chemistry Organic Chemistry Second Stage Lecture 1 ( B

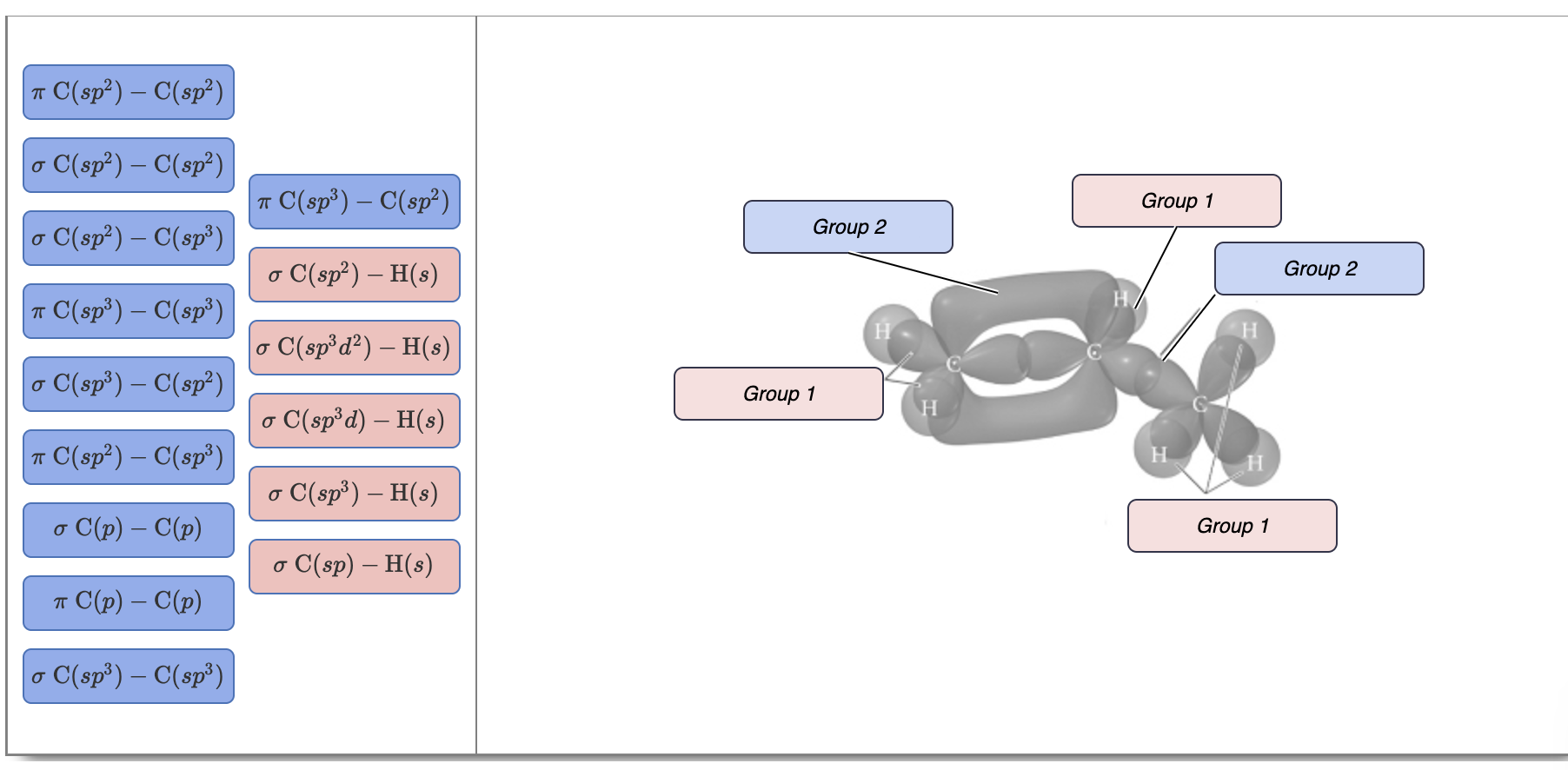
SOLVED: What atomic or hybrid orbitals make up the π bond between C1 and C2 in propylene, CH2CHCH3? π orbital on C1 π orbital on C2 How many bonds does C1 have
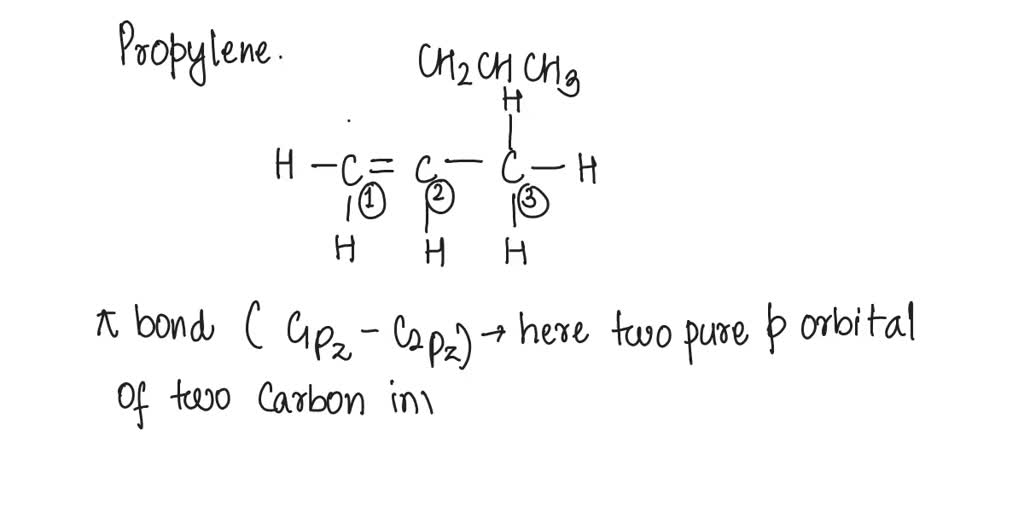
SOLVED: What atomic or hybrid orbitals make up the π bond between C1 and C2 in propylene, CH3(CHCH3)? (C1 is the first carbon in the formula as written: orbital on C1 orbital
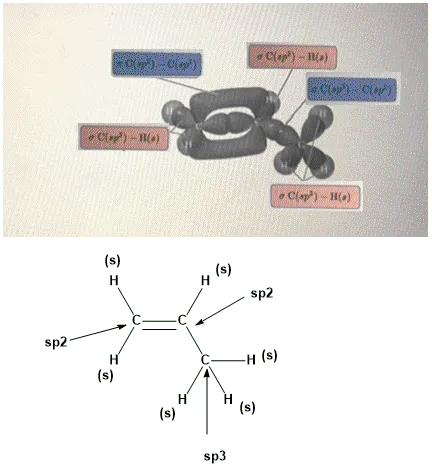
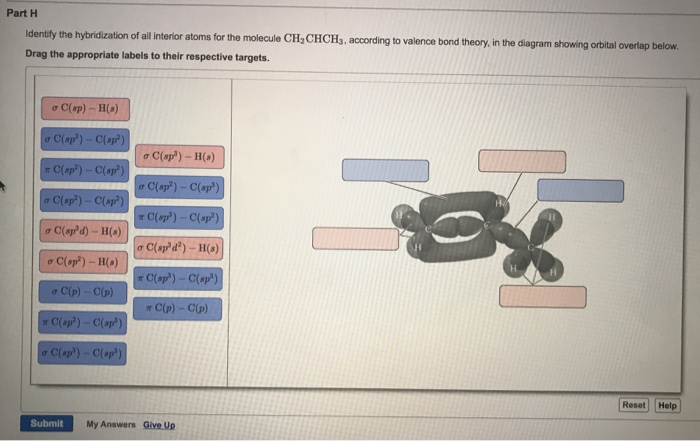
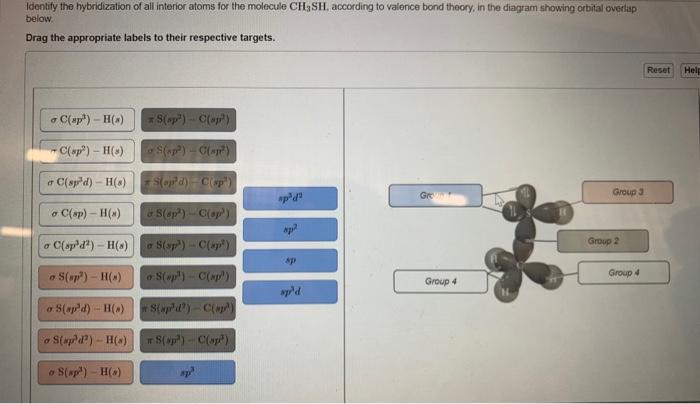
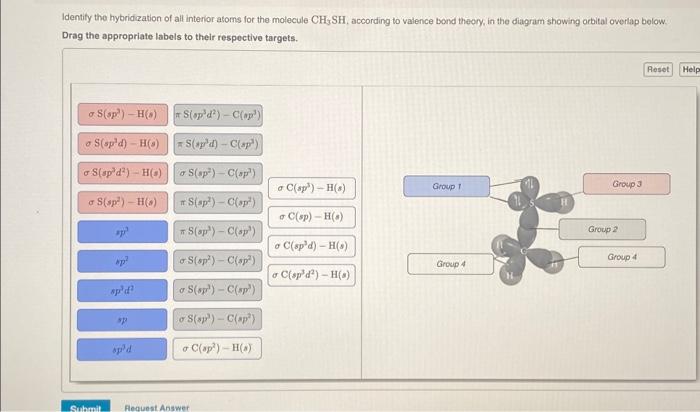
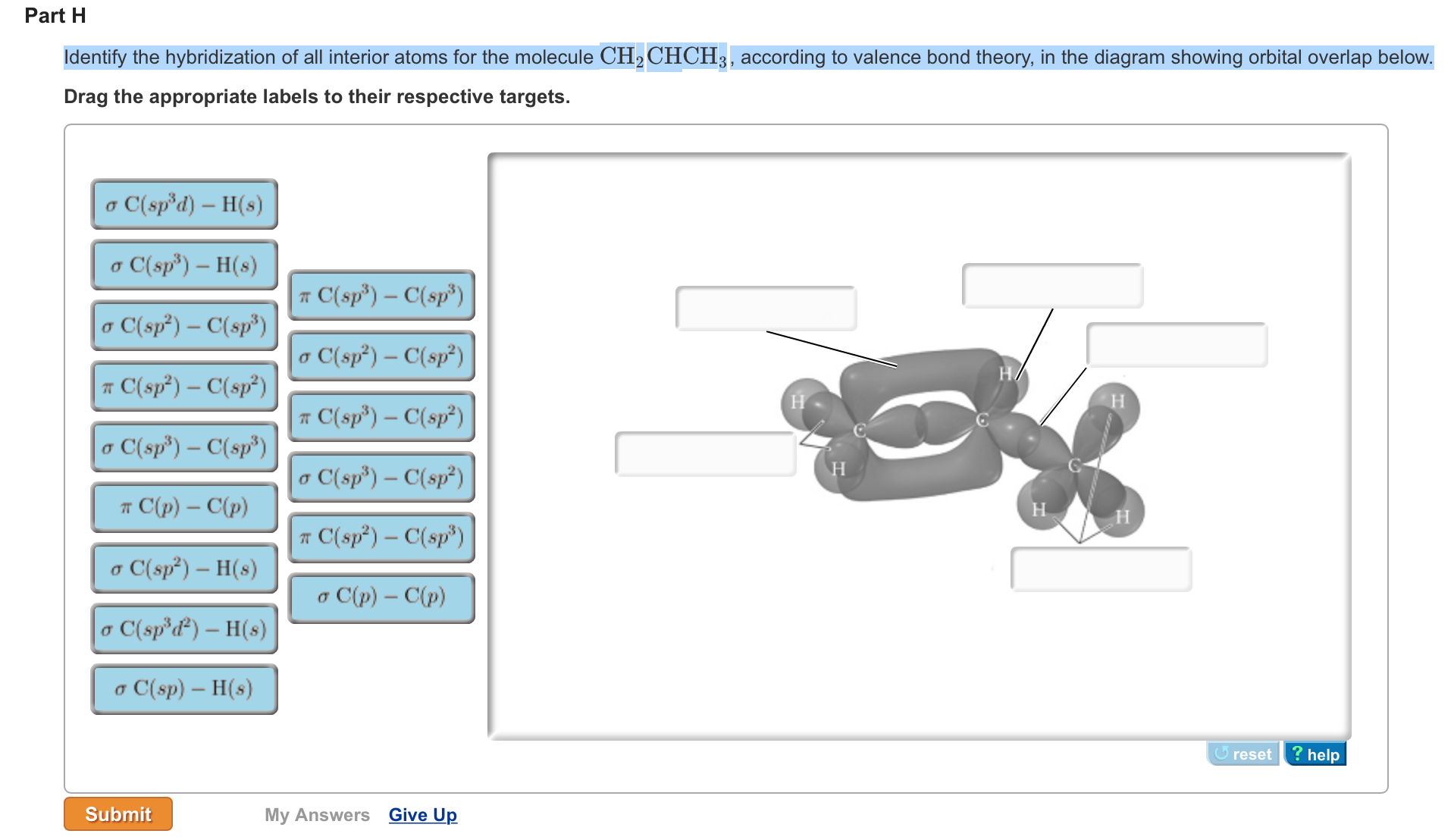
OneClass: Part H Identify the hybridization of all interior atoms for the molecule CH2 CHCHs, accordi...
Draw the Lewis structure for the molecule CH3CH2CCH. How many sigma and pi bonds does it contain? a) 11 sigma, 2 pi b) 9 sigma, 1 pi c) 8 sigma, 3 pi

SOLVED: Text: Propane; CH3CH2CH3 and propene, CH3CH=CH2 are members of different homologous series. (1) Draw diagrams to show how sigma (σ) and pi (π) bonds are formed between carbon atoms. (ii) State